ANZUP at ASCO GU 2026
Overview
ANZUP research was in the global spotlight at ASCO GU 2026 in San Francisco, with new findings from the CLIMATE study (ANZUP 1906) presented alongside poster presentations for the P3BEP (ANZUP 1302) and ENZAMET (ANZUP 1304) studies.
We also facilitated the international steering committee meetings (ITSC) for ENZAMET, ENZARAD, and DASL-HiCAP.
CLIMATE

New research findings were reported at ASCO GU 2026 from the CLIMATE study (ANZUP 1906), led by ANZUP in collaboration with the Walter and Eliza Hall Institute of Medical Research and Peter MacCallum Cancer Centre.
The findings reveal a simple blood test, measuring the levels of a highly sensitive and specific marker (miR-371) of testicular cancer germ cell tumours can help predict which men with early-stage testicular cancer are most likely to have their cancer return.
People diagnosed with stage 1 testicular germ cell tumour are often treated with surgery alone and then closely monitored through active surveillance. Some people with high-risk disease will be treated with chemotherapy to prevent relapse. While most men with early-stage testicular cancer have excellent long-term outcomes, around one in four people will see their cancer return within five years. This ongoing uncertainty, and the need to optimise therapy for those that are most likely to benefit, highlights the need to improve how an individuals risk of relapse is identified.
The CLIMATE study investigated a blood-based marker called miR-371, which is released into the bloodstream by testicular cancer cells. The study tested whether miR-371 could detect tiny amounts of residual cancer after surgery, specifically in those with testicular germ cell tumours, following the surgical removal of a testicle. The interim results showed that miR-371 performed better than the predictive tools that are currently available in predicting those people that are most likely to have their cancer return after initial treatment. With further validation, this test could add a valuable tool to inform the management of early-stage testicular cancer.
You can read more in our media release.
P3BEP

An international randomized phase 3 trial of accelerated versus standard BEP chemotherapy for individuals aged 11-50 years with intermediate and poor-risk metastatic germ cell tumours (GCTs) – poster presented by Associate Professor Ben Tran.
Germ cell tumours (GCTs) such as testicular cancer typically affect young adults. Around a third of patients still relapse despite best the available BEP (bleomycin; etoposide; cisplatin) chemotherapy.
P3BEP (ANZUP1302) is the largest randomised clinical trial exploring the possible benefits of accelerated BEP chemotherapy. In P3BEP, eligible participants are randomly assigned to receive either standard 3-weekly BEP or accelerated 2-weekly BEP. P3BEP opened in 2014 and is now available at 205 hospitals across Australia, New Zealand, United Kingdom and USA. This Trial-In-Progress poster for ASCO GU shows that P3BEP is steadily approaching its recruitment goal (407/500 participants), with the final analysis expected in 2029.
The results of P3BEP will provide robust evidence to support the use of accelerated BEP in the treatment of advanced GCTs, with the potential to change standard of care, avoid the need for costly salvage treatment, improve cure rates, and shorten the duration of intensive chemotherapy.
ENZAMET
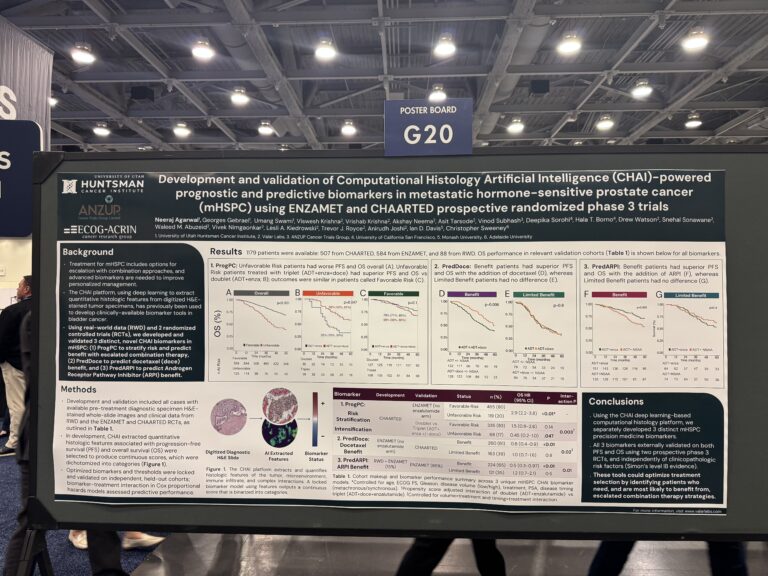
Development and validation of computational histology artificial intelligence (CHAI)-powered prognostic and predictive biomarkers in metastatic hormone-sensitive prostate cancer (mHSPC) using ENZAMET and CHAARTED prospective randomized phase 3 trials (RCT) – first author Neeraj Agarwal.
Clinicians managing people diagnosed with metastatic hormone-sensitive prostate cancer (mHSPC) often face a critical question at the start of treatment: which therapy (or combination of therapies) is most likely to maximise benefit and minimise harm. While advanced hormone therapies (ADT plus ARPI’s) plus or minus chemotherapy are effective, reliable tools to guide these choices are lacking. In collaboration with Valar Labs, this study developed a novel AI tool, CHAI (Computational Histology Artificial Intelligence), which can assess overall risk and predict who is most likely to benefit from each treatment. The CHAI algorithm was validated on two large international trials, including ANZUP’s practice-changing ENZAMET study and CHAARTED, and the results showed that CHAI could help optimise treatment selection by identifying those people who need, and are most likely to benefit from treatment intensification.
ANZUP International Trial Steering Committee (ITSC) Meetings

ANZUP was pleased to facilitate the international steering committee meetings for both ENZAMET and ENZARAD, as well as DASL-HiCAP, held in San Francisco last week.
Both hybrid meetings were highly productive, providing valuable opportunities for collaborators and international partners to strengthen relationships, explore the current GU clinical trials landscape, identify research gaps, discuss regional updates and challenges, and review ongoing and planned sub-studies.
Following the ITSC, we hosted a celebratory gathering to recognise ANZUP members and studies featured at ASCO GU 2026. We are incredibly proud of the contribution our research continues to make towards our vision of enabling people to live free from the fear of cancer. Thank you to everyone who attended, contributed and joined in the celebration!
